A bottom-up synthesis of a next-generation material, nanographene, using Pd(OAc)2/o-chloranil as a catalyst is developed.
Graphene, which consists of planar sheets of carbon atoms arranged in a honeycomb crystal lattice, has received tremendous attention in almost all areas of science and technology. Due to its interesting properties, particularly its high electron mobility (electron mobility indicates how fast an electron can move through a substrate), graphene may yield semiconductors with superior performances to current silicon semiconductors. Additionally, graphene may be used in high-efficiency solar panels. Thus, graphene is a next-generation material with numerous potential applications.
Nano-meter-sized (Nano meter is 10-9 meter) graphene is referred to as "nanographene" (Fig. 1). The properties of graphene are determined by the shape at nano meter level. Consequently, nanoscale shape-controlling technologies are necessary to realize practical applications of graphene. Currently, there are several methods to synthesize graphene such as the "Scotch tape method", which pulls single-atom layers from graphite. Unfortunately it is difficult to control the shape of nanographene using these methods. Hence, a bottom-up approach, which means to synthesize nanographene fragments from its "components" and bind these fragments, must be developed to resolve this issue. Although it may seem intuitive and simple to develop a bottom-up synthesis, to date, a bottom-up nanographene synthesis does not exist.
In order to realize bottom-up approach, Kenichiro Itami, and his research collaborator Katsuaki Kawasumi, Kenji Mochida, and Yasutomo Segawa from Elucidation and Design of Materials and Molecular Functions GCOE Program at Nagoya University have developed the "Pd(OAc)2/o-chloranil system" as a catalyst to synthesize nanographene fragments.
A catalyst, which enhances the chemical reaction and acts like "glue", is key factor of a bottom up approach. In this case, the catalytic Pd(OAc)2/o-chloranil system helps bind polycyclic aromatic hydrocarbons (PAHs) and arylboroxins, which are components of nanographene. A new bond is formed in a regioselective fashion via C?H/C?B cross-coupling. After C?H/C?B cross-coupling, the carbon sheets (Fig.2), which are nanographene fragments, are produced using FeCl3 as an oxidant (Fig.3). And it is able to construct nanographene from these fragments using a chemical or physical method.
In discussing the significance of their research, Itami explains, "Planning the bottom-up approach is easy, but realizing a method is much harder because it is difficult to discover a catalyst that "glues" compounds, which is a key factor in the bottom-up approach. We have accepted this challenge, and are the first group to successfully develop a catalyst to synthesize nanographene fragments. The proposed catalyst may be applicable to synthesize other extended PAHs. In the future, we intend to complete this bottom-up approach by constructing nanographene from the fragments using a chemical or physical method. Additionally, we will apply this catalyst to synthesize PAHs other than graphene. "
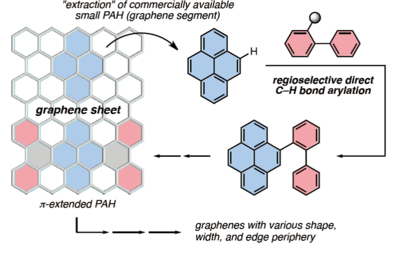
Fig. 1:Bottom-up approach for the synthesis of nanographene.
Blue and red denote PAH and phenylboron compounds, respectively.
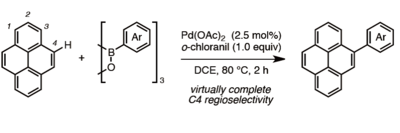
Fig. 2:C-H/C-B cross-coupling between pyrene (one of PAHs) and arylboroxin.
Red circle shows the binding position of pyrene.
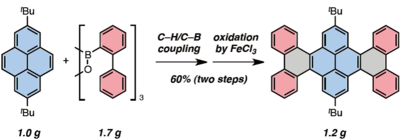
Fig. 3:After the C-H/C-B cross-coupling reaction, FeCl3 is used as an oxidant
to synthesize a nanographene fragment.

Fig. 4:Itami explain "Understanding the bottom-up approach is easy,
but realizing a method is much harder. We have accepted this challenge,
and are the first group to successfully develop a catalyst to synthesize
nano graphene fragments."

Fig. 5:Inside of Itami Laboratory.
Affiliated Researchers
Elucidation and Design of Materials and Molecular Functions GCOE Program
Reference
1. Kenji Mochida, Katsuaki Kawasumi, Yasutomo Segawa, Kenichiro Itami, "Direct Arylation of Polycyclic Aromatic Hydrocarbons through Palladium Catalysis", J. Am. Chem. Soc., 2011, 133 (28), pp. 10716-10719..